This is the title of a talk I gave on March 9, 2023, at a special Symposium of the Spring Meeting of the Section on Atomic, Molecular, and Optical Physics of the German Physical Society. The purpose of the Symposium at the University of Hannover was to honor and recognize over 50 years of work by a colleague of mine, Professor Eberhard Tiemann. Eberhard’s seminal work in molecular spectroscopy has been essential to a body of international research over the last 3 decades involving many fruitful research directions with ultracold matter. The field of molecular spectroscopy deals with the “spectrum” of discrete wavelengths (or “spectral lines”) absorbed or emitted by a specific molecule.
The false color but beautiful picture above shows anything but cold matter. Rather it shows a recent NASA image of the Sun taken at the deep-red 656-nanometer wavelength of the H-Alpha line in the Balmer series of emission lines of atomic hydrogen. Such images help us understand the structure and properties of the Sun. Its surface is composed primarily of Hydrogen at a temperature of around 5500 degrees Celsius. In the 19th Century, scientists discovered that various hot gases would only emit light with very specific wavelengths. Why lines existed at discrete wavelengths was a major scientific puzzle, unexplainable by the physics of the time. Johann Jakob Balmer (1825-1898) was a math and Latin teacher at a girl’s school in Basel, Switzerland, and in 1885 published a paper noting that the specific wavelengths of a series of known lines emitted by hot hydrogen gas fit to a very simple mathematical formula (and later more lines came to be added to the series). The H-Alpha line in the above contemporary picture of the Sun is the first line in Balmer’s series. Swedish physicist Joseph Rydberg generalized the formula in 1888 to incorporate more Hydrogen lines.
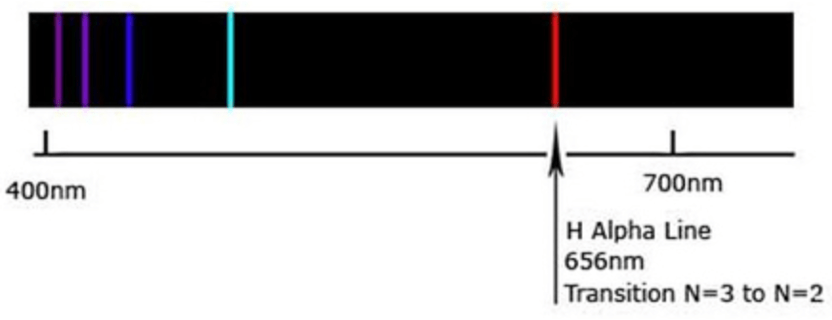
In the early 20th Century Niels Bohr developed his famous “quantum” model of the Hydrogen atom that could quantitatively explain the formulas of Balmer and Rydberg. Hydrogen is the simplest atom, comprised of a heavy positively charged proton and a light negatively charged electron. In Bohr’s theory, the electron could only orbit the proton in discrete orbits, drawing upon Max Planck’s discovery in 1900 of the quantum of energy associated with light. This is what Bohr said in his 1913 paper: “Whatever the alteration in the laws of motion of the electrons may be, it seems necessary to introduce in the laws in question a quantity foreign to the classical electrodynamics, i. e. Planck’s constant, or as it often is called the elementary quantum of action.” Planck and Bohr knew that the “classical electrodynamics” of particles and light from Newton and Maxwell was incapable of explaining the observed facts about atomic spectra. The principle Bohr used was to invoke only “orbits” that had very specific integer multiples of rotational angular momentum measured in suitable units of Planck’s quantization constant introduced in 1900. The Balmer H-Alpha line corresponds to a “quantum jump” between the orbit with integer 3 to the one with integer 2. Given the known value of Planck’s constant, the wavelength had to be 656 nanometers. This may sound very abstract, mathematical, and obscure, but something new was afoot and Bohr was on the right trail. While his atomic theory is now known to be obsolete and has been supplanted by the modern quantum theory of the atom, it got the basics right and proved to be an important step in understanding what was needed in the development of the proper Quantum Theory of matter.
The Quantum Theory, despite its many counter-intuitive features, has proved to be incredibly successful in explaining matter at its most “fundamental” level of particles, atoms, molecules, and photons. It accounts for the very complex set of spectral lines that serve as unique “fingerprints” that identify every specific kind of atom or molecule in the universe. The Quantum Theory gives a powerfully unifying theory across many branches of science and technology. This unifying beauty is in addition to an intrinsic mathematical beauty of the theory. Quantum Theory is our key to understanding the cosmos. The picture of the Sun reminds us that practically everything we know about the cosmos beyond the earth comes to us by way of light [1], which can be analyzed by various kinds of “spectrometers” into its individual wavelength components. For example, we have not only visible light spectroscopy, but microwave, infrared, ultraviolet, x-ray, and gamma-ray spectroscopy, all different forms of “light.” This is how we know about the composition of distant stars, planetary atmospheres, interstellar molecular clouds, the motion of distant galaxies, the expansion of the universe, and mysterious objects like “black holes” or neutron stars [2].
Professor Tiemann and his colleagues have spent countless hours analyzing thousands of spectral lines of dozens of different molecular species comprised of two atoms. The fruit of their labors are sets of several dozen numbers (“parameters”) that fully specify the quantum mechanical forces between the two atoms that are needed to calculate all the individual quantum states associated with the molecule formed from two atoms each in its quantum state of lowest energy. That is what I do—these kinds of calculations. In this way, it is possible to calculate from first principles and standard mathematics the needed properties to fully characterize the collisions of two ultracold atoms and the formation from them of a two-atom molecule in any desired quantum state. It is now routine to produce in the laboratory samples of certain kinds of atoms at the incredibly low temperature of 1 microkelvin or less (this is nearly a billion times colder than room temperature). Professor Tiemann’s work was invaluable in the actual production in 2008 of Potassium-Rubidium (KRb) molecules in their lowest energy state with a temperature of 350 nanokelvins by my colleagues at the NIST/JILA laboratories of the University of Colorado, Boulder. I told the story of this work (and more) in my recent talk [3]. The story started with a joint paper I had done with Eberhard when I was visiting Hannover in 2002, proposing such an experiment, with the goal of making a quantum superfluid of ultracold molecules.
This brings up a final matter relating to the unity and beauty of physics. Scientists can now make many different species of atoms or molecules at such incredibly low temperatures, at which the quantum properties of their motion become clearly manifest. We can not only make cold gases but also optical “lattices” to hold regular arrays of individual atoms or molecules or even arrays of optical “tweezers” that can be moved around at will and that each contains a single atom or molecule. In these various ways, the quantum properties of complex condensed matter systems like superfluids can be probed and studied. Because the equations of physics scale across vast magnitudes of energy or density, tiny ensembles of cold atoms can even simulate a variety of phenomena in condensed matter physics and even explore the properties of large-scale structures in the universe. For example, the quantum “equation of state” that can be tested with tiny ensembles of cold atoms even helps us to understand the properties of quantum matter in the interior of neutron stars. We might even envision how someday single atoms or molecules arranged in lattices or tweezer arrays [4] might be used as “quantum bits” in a “quantum computer” that, at least with respect to certain tasks, could outperform any possible contemporary “classical” computer made from conventional computer chips.
Well, there is a lot I could talk about, but that exceeds anything I can include in this short post. So more will have to wait until later. But I hope I have scratched the surface of the unity, power, and beauty of what I see in the ultimately simple concepts of quantum physics to characterize the universe in which we live across vast scales of size and complexity, from the atomic to the cosmological. That covers a lot of territory. But perhaps the most amazing feature of our universe is the human mind, which takes all this in and renders it intelligible. Quantum mechanics is very intelligible, in spite of the fact that no one yet can specify how it should be interpreted in “classical” terms (Bohr’s point). Perhaps the “quantum world” is so strange and different from our “ordinary, everyday” world that there must be aspects of it that forever exceed our capacity to articulate, except in formal, mathematical terms.
Notes:
1. An obvious exception is rocket probes of the Moon or Mars, which can work with local rock samples, for example.
2. More recently, the first “non-light” kind of observation of distant objects in the universe has come about through the success of gravitational wave astronomy. Interestingly, it is now an observed fact that gravitational waves travel at the speed of light, the upper “speed limit” in the universe, in spite of the fact that gravity is not light.
3. Download the Dropbox file and view it in Powerpoint if you want it to display with the correct original font.
4. See, for example, the review article by Adam M. Kaufman and Kang-Kuen Ni, “Quantum science with optical tweezer arrays of ultracold atoms and molecules,” Nature Physics, Vol. 17, p. 1324 (2021) (the article file is also available at this open link.)


