(This post was first published on the BioLogos.org web site March 17, 2014. It is also available on the BioLogos site in a reformatted version.)
Before there ever could be biomolecules, or a genome, or living beings, there had to be carbon and the other chemical elements that are essential to life. The science of carbon is remarkable, and the more one knows about it, the more one can stand in awe of the amazing universe in which we live. My career in physics—in particular, the quantum physics of atoms and molecules and light at the interface of chemistry and physics—has taught me the depth and power of the natural sciences to understand the world. It is a pleasure to be able to contribute to the Biologos blog a few thoughts about carbon: how it came to be made in the fire of the stars of the early universe and how it enables the remarkable chemistry of life written out in the words of the genome. Putting it all together draws on connections between atomic and nuclear physics, cosmology, quantum theory, chemistry, biology, and what science is all about in the first place.
I tell the story based on all the positive knowledge we have from the sciences. Does it have anything to do with God and humanity? Read to the end of Part 2 and decide for yourself. First, let us take a whirlwind tour of the picture science gives us of the early universe and of the origin of the chemical elements.
According to the best current measurements, our universe is approximately 13.8 billion years old, and had a long history before there was life on earth. After an initial “Big Bang,” the universe rapidly expanded and cooled so that after a few minutes the present abundance of most of the atomic nuclei in the universe had been established, about ¾ hydrogen and ¼ helium, plus a trace of lithium. The simplest atomic nucleus is hydrogen, 1H, having a single positively charged proton. The helium nucleus, 4He, is known as an alpha particle, comprised of two protons and two neutrons. In this early stage of the universe, there were no nuclei of species heavier than 7Li (lithium with 3 protons and 4 neutrons) such as carbon, oxygen, or iron.
After about 380,000 years of expansion and cooling, the positively charged hydrogen and helium nuclei recombined with negatively charged electrons to make ordinary electrically neutral hydrogen and helium atoms. The universe was still mostly uniform without clumping into galaxies and stars, but once it was composed primarily of neutral atoms, it became transparent to light, that is, light could propagate freely throughout the universe. This light has continued to cool, and its afterglow is known as the microwave cosmic background radiation, which radiates from every direction in the sky, providing prime evidence of Big Bang cosmology and giving a measure of the age of the universe.
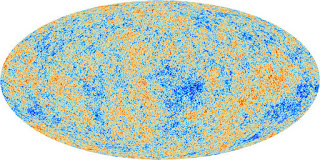
What about the heavier elements? Since stable nuclei heavier than lithium didn’t exist in the very early stages of the universe, where did they come from? How were they built up?
After the separation of light and matter in the early universe, the hydrogen and helium began to clump into large clouds of gas that under the influence of gravity condensed into galaxies and stars. The first stars and galaxies had already formed by the time the universe was one billion years old. It turns out that the heavier elements can be made in the hot interior of stars by fusing together lighter nuclei via sequences of nuclear reactions that can explain the observed abundance of these elements. It is only in the dying phase of certain types of stars that temperature and pressure is sufficiently high that these fusion processes occur to make the heavier elements. These elements are then expelled into the surrounding interstellar medium by the exploding star at the end of its life. The clouds of gas formed this way later condense into new stars, such as our sun, some of which have accompanying planetary systems. Consequently, before there could ever be carbon, there had to be a first generation of stars to be born and die. In other words, given what we understand about the laws of nature and star formation and evolution, the universe actually needs to be billions of years old before carbon-based life could be present.
How the heavier elements are made in stars was worked out in the 1940s and 1950s through discoveries about nuclear physics and nuclear reactions. A classic paper published in 1957, “Synthesis of the Elements in Stars,” by Margaret and Geoffery Burbidge, William Fowler, and Fred Hoyle, laid out the basic framework that remains with us today. Fowler received the 1983 Nobel Prize in Physics for his work on nucleosynthesis, the two Burbidges received the Gold Medal of the Royal Astronomical Society in 2005, and Hoyle was later knighted for his work in astrophysics and was awarded the prestigious Crafoord Prize of the Swedish Academy of Sciences in 1997 for his work on the formation of the elements in stars.
Getting the heavier elements requires first making a carbon nucleus, which is very difficult. Making 12C requires that three alpha particles, 4He, fuse together. This is called the triple-alpha process, but it is impossible at the 15 milliondegree temperature inside a normal star like our sun, because the average velocity of the alpha particles is too low for them to overcome the very strong repulsive electric forces between the positively charged 4He nuclei. Hans Bethe had already shown in 1939 that a temperature of 1 billion degrees would be required for such repulsion to be overcome. But such a high temperature does not occur even in stars.

Fred Hoyle in the 1950s.
In 1953 the young astrophysicist Fred Hoyle realized that accounting for the relative abundances of carbon and oxygen in the universe required that there be a special quantum state of the 12C nucleus that would allow it to form in stars at temperatures around only 100 million degrees. Such high temperatures are only possible in the interior of a star that is collapsing under the tremendous force of gravity after its hydrogen fuel is nearly spent. The postulated quantum state, which may or may not exist, had to have just the right properties to allow fast enough production of 12C nuclei but to prevent their destruction by rapid conversion to 16O upon fusing with another alpha particle. While visiting the Kellogg Radiation Laboratory at Caltech, Hoyle told William Fowler and his colleagues and students there about his prediction, and it was verified through laboratory experiments that the needed state existed at close to the predicted value. With this knowledge in hand, Hoyle and others could then understand how the heavier elements could be made through sequences of nuclear reactions starting with 12C and 16O, and the foundation was laid for understanding how all the heavier elements came to be.
All the elements needed for life are synthesized in the late stages of the life cycle of certain stars. Without the Hoyle state in the triple alpha process, we would not be here as living beings who can understand such things. In an article entitled “The Universe: Past and Present Reflections,” published in the Annual Reviews of Astronomy and Astrophysics in 1983, Hoyle wrote the following (Vol. 20, p. 16):
“From 1953 onward, Willy Fowler and I have always been intrigued by the remarkable relation of the 7.65 Mev energy level in the nucleus of 12C to the 7.12 Mev level in 16O. If you wanted to produce carbon and oxygen in roughly equal quantities in stellar nucleosynthesis, these are the two levels you would have to fix, and your fixing would have to be just where these levels are actually found to be. Another put up job? Following the above argument, I am inclined to think so. A common sense interpretation of the facts suggests that a superintellect has monkeyed with physics, as well as with chemistry and biology, and that there are no blind forces worth speaking of in nature.”
Hoyle was known for making controversial claims. While few scientists would claim that the science would establish that “a superintellect has monkeyed with physics,” the Hoyle state does provide another example where the laws of physics of our actual universe are fine tuned such that carbon-based life is possible.
Be sure to check out the next part to learn more about the intersection of science, carbon, and life.
